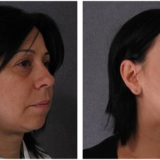
Phentermine and Topamax (or Topiramate), together sold under the trade name Qsymia, is a highly popular combination medication for obesity and weight loss. While the former is basically an appetite suppressant and a metabolism stimulant for the obese, the latter is an anti-convulsant with certain weight loss side effects. Taken orally as capsules, the medication is approved in the USA for use by adults having weight loss issues and a very high body mass index. Though as a prescription pill, any variation in the prescribed dosage is not recommended, Qsymia is also marketed over the counter without prescription, as well as online with world-wide delivery options. Topamax is an anti-obesity drug that has passed muster with the drug authorities.
How does it work?
The pill is a recent introduction into the slimmer’s world. Backed with government approvals, its growing popularity as a weight loss product is attributed toits stated effectiveness in helping people lose weight andcurb one’s appetite, while simultaneously burning and suppressing further production of body fat without any negative effects whatsoever on one’s energy levels and moods. Clinical trials have shownproof of the beneficial effect of Qsymia when the medication is combined with exercise and diet restrictions. Click here to know the clinical results for Topamax .
Beware of its potential risks
However, this is not to say that the intake of Qsymia is not accompanied by any potential risk or contra-indications. On the contrary, these hazards are many and alarmingly serious too. Dizzinessand gastro intestinal side effects such as dry mouth, vomiting, diarrhea, constipation and nausea are cautioned as common side effects. The drug label carries a warning against blood pressure, pulmonary hypertension and other adverse cardio vascular conditions that may result as adverse reactions. Neurologically, continued usage of the drug could produce depression, tremors and withdrawal symptoms and even similar to addiction in addition to cognitive impairment. Abstinence from alcohol is necessary while taking Qsymia. The drug is not advised for glaucoma, heart or kidney patients or those with a history of epilepsy, nor to pregnant or lactating mothers. Fetal toxicity, if taken during pregnancy has been reported. The list of restrictions certainly does seem to suggest very careful and cautious usage.
Why then is it gaining acceptance?
Curing the overweight through anorexia would, prima facie, appear confusing and questionable. Yet, this is what Qsymia professes to do. Combined with the potentially harmful health conditions and contra indications cited due to its usage, it would appear that the potential health issues that might arise due to this medication far outweigh its benefits. Yet, Qsymiahas seen a growing popularity and usage – largely since it is popularly marketed as a primarily plant- based supplement,that is also easily available online and over-the-counter;and since it guarantees accelerated weight loss without the compulsions or associate need for strenuousexercise. Further medical opinion holds, considering the many side effects that might result, that in addition to a prescribing physician, the drug should be prescribed under the guidance of a team of doctors who would include a dietitian and an obesity specialist.